
Effects of Ultrafine Powderization and Hydrogen-Rich Water Extraction on Antioxidant Activity of Red Ginseng
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
The health benefits of red ginseng are attributed to its antioxidant activity. Current information regarding the effects of ultrafine powdering and hydrogen-rich water extraction on the antioxidant activity of red ginseng is limited.
Particle sizes, hydration properties, and antioxidant activity of red ginseng were investigated to determine its physicochemical characteristics. After ultrafine powdering, the particle size of the ultrafine red ginseng powder was reduced 2.5-fold and the water solubility index was increased by 7% compared to that of coarse red ginseng powder. Notably, the antioxidant activity of ultrafine red ginseng powder was increased as the water solubility index increased (p < 0.05). When ultrafine red ginseng powder was extracted with hydrogen-rich water, the total phenolic and flavonoid contents increased significantly (p < 0.05) because molecular hydrogen protected it from destruction by oxidation. Therefore, the antioxidant activity also increased significantly after hydrogen-rich water extraction (p < 0.05).
This study demonstrated that the antioxidant activity of red ginseng can be increased by ultrafine powdering and hydrogen-rich water extraction.
Keywords:
Red Ginseng, Ultrafine Powderization, Hydrogen-Rich Water Extraction, Antioxidant Activity, Hydration PropertyINTRODUCTION
Ginseng, the root of Panax ginseng C. A. Meyer, is referred to as “the king of herbs” and literally means “the essence of human” (Park et al., 2006). For thousands of years, people have used ginseng as a dietary supplement, functional food, and traditional medicine (Williamson et al., 2020) to improve vital energy and preserve bodily homeostasis (Choi, 2008). The biological and pharmacological properties of ginseng include anti-inflammatory, anti-stress, anti-aging, antidiabetic, and antioxidant properties. Furthermore, ginseng has an effect in cancer prevention, arteriosclerosis, hypertension, immune system enhancement, brain function restoration, and fatigue alleviation (Lee et al., 2010). One of the mechanisms in health-related benefits may be related to its antioxidant properties (Kim et al., 2011).
Depending on its processing method, ginseng is categorized as fresh ginseng, white ginseng (WG), and red ginseng (RG) (Matsuura et al., 1994; Nam, 2005). The dried ginseng root is called WG, while the ginseng root that has been steamed at around 100oC and dried is called RG. Compared to WG, RG may be kept for longer periods of time (Jeong et al., 1997; Jeon et al., 2005; Seo et al., 2013). RG has been utilized as a valuable medicinal element because its pharmacological actions are improved more than those of WG (Kim et al., 2000; Nam, 2005). The change in chemical components by steaming enhances the biological activities of RG (Yun et al., 1983; Kim et al., 1996; Kim et al., 2000). It has already been documented that the steamed ginseng roots exhibit antioxidant activity (Yang et al., 2006; Kim et al., 2007).
Phytochemicals can be free, chemically cross-linked, or physically entrapped within plant cell wall matrix. Free phytochemicals are generally in vacuole, whereas entrapped and bound phytochemicals are in the plant cell wall matrix (Gulsunoglu et al., 2019). The majority of phytochemicals are in the plant cell wall matrix. Pretreatment is required to release them from the plant cell wall matrix. Depending on the structure of plant cell wall matrix and the kinds of phytochemicals, this step may involve a variety of physicochemical treatments, including drying, grinding, sonication, and so on (Azwanida, 2015). Ultrafine powderization has been extensively researched in the field of grinding (Yun and Lee, 1994a, b; Yun et al., 1996). Ultrafine powderization increases surface area by decreasing particle size and destroys plant cell wall matrix. As a result, phytochemicals are more readily dissolved out from plant cell wall matrix (Hermansson, 1982; Yu et al., 2002a, b).
One or more hydroxyl groups (-OH) are joined to an aromatic ring in phenolic compounds. When oxygen or other oxidizing agents are present, these hydroxyl groups are vulnerable to oxidation processes (Waterhouse et al., 2006). Molecular hydrogen (MH) is a strong antioxidant. Consequently, it can protect phenolic compounds from oxidation reactions, making them less susceptible to breakdown (Hu et al., 2021). Therefore, during the extraction process, the phytochemicals may be protected from oxidation reactions by MH in water.
Very limited information is available on the effects of ultrafine powderization and hydrogen-rich water extraction on the antioxidant activity of RG. In this study, we investigated them and then discussed what factors may influence.
MATERIALS AND METHODS
1. Preparation of ginseng powder
Five-year-old fresh ginseng was purchased from a local market in Yeongju, Korea, and washed with tap water. WG was produced by drying fresh ginseng at 50℃ for 3 days, using a dry oven (WFO-700W: Tokyo Rikakikai Co., Ltd., Tokyo, Japan). RG was produced by steaming fresh ginseng with distilled water for 7 hours, using OCOO (OC-S1170S; OCOO Co., Ltd., Seoul, Korea) and then drying at 50℃ for 3 days.
2. Coarse and ultrafine powderization
WG and RG were ground to coarse powder, using a food mixer (SHMF-3500SS; Hanil Electronics Co., Ltd., Seoul, Korea). Red ginseng ultrafine (RGU) powder was produced by grinding red ginseng coarse (RGC) powder, using a low-temperature turbo mill (LS-001; Mechano Korea Co., Ltd., Chuncheon, Korea). The low-temperature turbo mill has a high physical power (impact, compression, and shear strength). The speed of rotor was 10,500 rpm. The temperature of mill chamber was kept at -18℃, and the temperature of ground products was kept at 25℃ to 30℃ by a cooling device using R-22. Also, the classification of ground powder was performed at the same time with milling by the principle of centrifugal and drag force.
3. Particle size analysis
The particle sizes of RGC and RGU powder were analyzed in triplicate using a Laser Diffraction Particle Size Analyzer (Bettersizer 260; Bettersize Inc., Costa Mesa, CA, USA). Powder was dispersed in distilled water before measurements. D50 is a median particle diameter, which is an equivalent volume diameter at 50% cumulative volume.
4. Powder morphology
The morphologies of RGC and RGU powder were observed, using a microscope (DM IL LED Fluo; Leica Microsystems CMS GMBH, Wetzlar, Germany). RGC and RGU powder were observed at 10× magnification.
5. Water absorption capacity, water solubility index, and swelling power
Water absorption capacity (WAC), water solubility index (WSI), and swelling power (SP) were determined in triplicate using the method of Anderson et al. (1969). One gram of sample was suspended in 20 mL of distilled water at room temperature for 30 min, gently stirring during this period, and then centrifuged at 2,000 × g (14,520 rpm) for 10 min. The supernatant was decanted into an evaporating dish of known weight. WAC, WSI, and SP were calculated by the following equations:
6. Extraction
We purchased 99.99% pure H2 gas from Dong-A Industrial Gas in Suwon, Korea, HPLC-grade solvents from Thermo Fisher Scientific, located in Waltham, MA, USA, and other reagent-grade chemicals from Daejung Chemical Co., in Seoul, Korea.
Hydrogen-rich water was prepared as follows. A 100 mL bottle was filled with 10 mL of distilled water, and the container was sealed with an aluminum lid and rubber stopper. Two needles were used to puncture the rubber stopper; one was positioned below water surface, while the other did not come into contact with water. For three minutes, H2 gas was bubbled via the needle that touched the water at a rate of 1 L/min. Both needles were removed from the rubber stopper at the same time after 3 min.
One gram of RGU powder was added to 10 mL of HPLC-grade distilled water or hydrogen-rich water, and then vortexed. Each sample was sonicated at 35℃ for 30 min using a POWERSONIC Ultrasonic Cleaner (POWERSONIC 420; Hwashin Tech Co., Ltd., Seoul, Korea). The mixture was filtered, using a syringe filter PTFE-H (SH25P045NL; Hyundai Micro Co., Ltd., Seoul, Korea). Then, the sample was centrifuged at 10,000 × g for 20 min, and the supernatant was used for antioxidant activity analysis.
7. Determination of total phenolic content
Total phenolic content (TPC) was determined in triplicate with a slight modification of the Folin-Denis method (Folin and Denis, 1912). The prepared extract of 0.5 mL was added to 0.5 mL of 1 N phenol reagent (Sigma-Aldrich Co., Saint Louis, MO, USA) and reacted at room temperature for 5 min, and then 1 mL of 10% sodium carbonate was added. The vortexed mixture was incubated at room temperature under dark conditions for 30 min. Absorbance values were measured at 725 nm, using a UV spectrophotometer (U-2001; Hitachi Ltd., Tokyo, Japan). TPC was expressed as gallic acid equivalent (mgGAE/g) on dry weight (DW).
8. Determination of total flavonoid content
Total flavonoid content (TFC) was determined in triplicate with a slight modification of the Moreno method (Moreno et al., 2000). The prepared extract of 0.1 mL was placed in a test tube, and 5 mL of 10% aluminum nitrate solution and 0.1 mL of 1 M potassium acetate solution were added. A total volume of 4 mL was made, adding 3.3 mL of tertiary distilled water. The vortexed mixture was incubated at room temperature under dark condition for 30 min. Absorbance values were measured at 415 nm, using a UV spectrophotometer (U-2001; Hitachi Ltd., Tokyo, Japan). TFC was expressed as quercetin equivalent (mgQE/g).
9. DPPH radical scavenging activity assay
DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity was determined in triplicate by Blois' method with a slight modification (Oh et al., 2015; Ka et al., 2016). The prepared extract of 1 mL was added to 3 mL of DPPH (Sigma-Aldrich Co., Ltd., Saint Louis, MO, USA) reagent. The vortexed mixture was incubated at room temperature under dark conditions for 30 min. Absorbance values were measured at 517 nm using a UV spectrophotometer (U-2001; Hitachi Ltd., Tokyo, Japan). DPPH radical scavenging activity was calculated according to the following equation:
10. ABTS radical scavenging activity assay
ABTS (2,2'-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) radical scavenging activity was determined in triplicate by the method described by Re et al. (1999) with a slight modification. ABTS radicals were produced by reacting 7 mM ABTS stock solution with 2.45 mM potassium persulfate and allowing the mixture to stand at room temperature under dark condition for 12 hr before use. The mixture was diluted with ethanol to get an absorbance of 0.700 ± 0.02 at 734 nm. The prepared extract of 0.2 mL was mixed with 3.8 mL of diluted solution, and absorbance values were determined at 734 nm using a spectrophotometer (U-2001; Hitachi Ltd., Tokyo, Japan) after 6 min of incubation. ABTS radical scavenging activity was calculated according to the following equation:
11. Statistical analysis
Data from all experiments were statistically analyzed via ANOVA and Duncan’s multiple range test, using SPSS software program (SPSS Inc., Chicago, IL, USA). Other data were analyzed statistically by independent-paired t-test, using the SPSS software (SPSS Inc., Chicago, IL, USA). A p value < 0.05 was considered significant.
RESULTS AND DISCUSSION
1. Properties of red ginseng powders
Cryogenic grinding, which was used in the ultrafine powderization of RGC powder, is a dry milling process to grind at low temperatures. It increases hardness and decreases the extensibility of ginseng cell wall matrix, thus making it brittle, which causes particle size to decrease quickly (Lee et al., 2012). The conditions of low-temperature turbo mill to produce RGU powder are described in Table 1. The photographs of WGC, RGC, and RGU powder are shown in Fig. 1. The effect of ultrafine powderization on particle size distribution is shown in Table 2. D50 of RGC powder was 195 μm, and that of RGU powder was 79 μm. In another study, the particle size of RGU powder was similar to the particle size in this study (Kim et al., 2018). D50 of RGU powder was abruptly decreased. There was 2.5 times reduction in the particle size of RGU powder, compared to that of RGC powder.

Photographs of (A) white ginseng coarse, (B) red ginseng coarse, and (C) red ginseng ultrafine powder.
The surface areas of RGC and RGU powder were 17 m2/cm3 and 62 m2/cm3, respectively. The RGU powder containing relatively small particles had a larger surface area than the RGC powder containing relatively large particles. This was in a good agreement with the results of the particle size distributions of RGC and RGU powder. Ultrafine powderization decreased particle size distribution toward small size and produced ultrafine powder with smaller particles. In general, the surface area of powder with smaller particles is larger than that of powder with larger particles (Kim et al., 2018).
RGC and RGU powder were observed by a microscope (DM IL LED Fluo; Leica Microsystems CMS GMBH, Wetzlar, Germany). The changes in the structures of RGC and RGU powder are illustrated in Fig. 2. The microscopic images demonstrated that the particle size of RGU powder was decreased, compared to that of RGC powder. The microscopic image of RGC powder showed a mixture of large and small particles, whereas that of RGU powder showed uniform particle sizes. The particle size of RGC powder was considerably larger than that of RGU powder. Smaller particles that seemed to have broken off from larger particles indicated the impact of ultrafine powderization on RGC powder. According to particle sizes, the appearance of RGC powder was rougher because particle size was large. The color of RGC powder, in which particle size is larger, was darker than that of RGU powder, as shown in Fig. 1.
2. Effect of ultrafine powderization on hydration properties of red ginseng powders
The hydration properties of RGC and RGU powder are shown in Table 3, and Fig. 3, Fig. 4, and Fig. 5. In RGC powder, WAC and SP were decreased after steaming, compared to those of WGC powder. WAC and SP of RGC powder were 5.0 g/g and 2.0, respectively, and those of WGC powder were 7.3 g/g and 2.7, respectively. WSI, which is often used as an indicator of the degradation of molecular components (Kirby et al., 1988), measures the amount of soluble components released from plant materials. The result showed that WSI was increased by steaming. WSI was 39.3% in RGC powder and 36.9% in WGC powder. WSI of RGC powder was increased by 7%, compared to that of WGC powder.

Water absorption capacity, water solubility index, and swelling power of white and red ginseng powders.
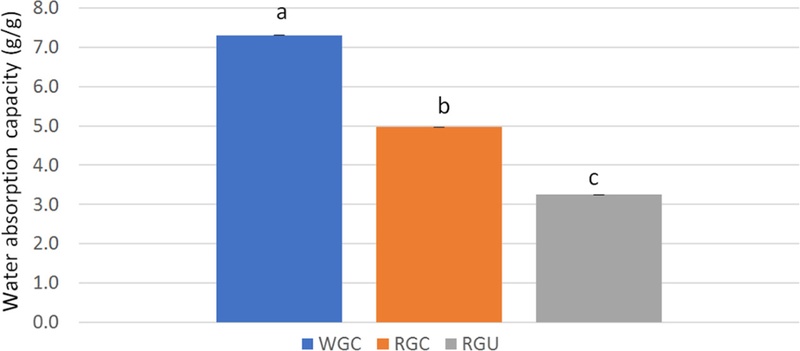
Water absorption capacity of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder.
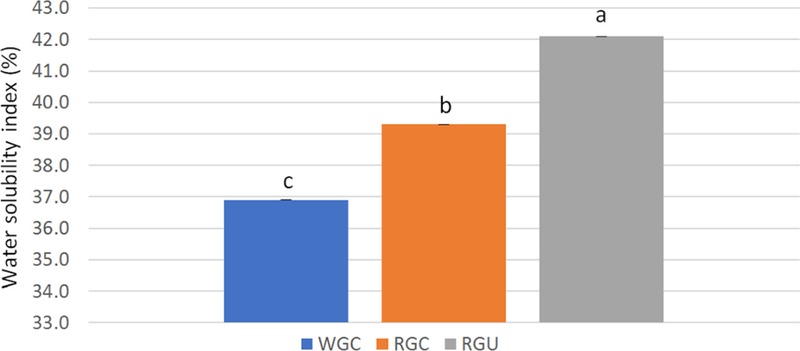
Water solubility index of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder.
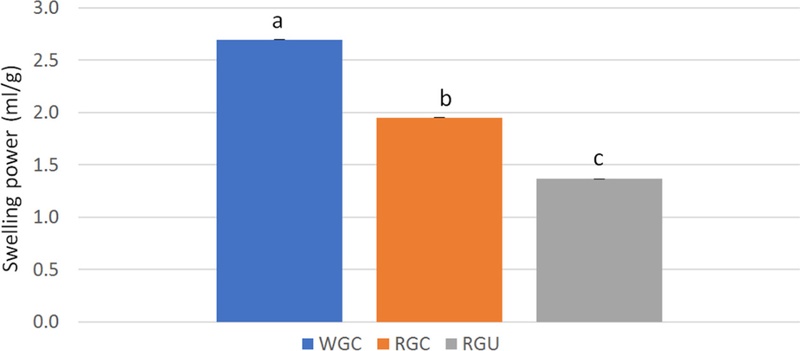
Swelling power of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). Abbreviations: WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder
After ultrafine powderization, the WAC and SP of RGU powder were 3.2 g/g and 1.4, respectively. WAC and SP of RGU powder were decreased by 35% and 30%, respectively, compared to those of RGC powder. However, the WSI of RGU powder with the D50 of 79 μm was 42.1%. WSI of RGU powder was increased by 7%, compared with that of RGC powder. WSI of RGU powder was found to be significantly higher than that of RGC powder (p < 0.05). Other studies had shown similar results that when using wheat bran and wheat straw powder, WAC was decreased in ultrafine powder (Rosa et al., 2015; Yang et al., 2014), and when using white ginseng powder, SP was decreased and WSI was increased in ultrafine powder (Lee et al., 2013).
If ginseng is steamed, the physical structure of ginseng is affected, and so the hydration properties of ginseng, such as WAC, WSI, and SP, are changed. WAC is related to the surface area and porous structure of plant material, which promote the bonding of water molecules to hydrophilic groups. After steaming, the porous structure of plant material may be destroyed and surface area may be reduced, both leading to a decrease in WAC (Li et al., 2019).
SP of RGC powder was decreased with steaming, compared to that of WGC powder. The original structure of ginseng cell wall matrix was destroyed, and the amount of macromolecules was reduced, both leading to decrease in SP (Li et al., 2019).
Unlike WAC and SP decreased, the WSI of RGC powder was increased. It may be due to the fact that steaming loosens the tight structure of ginseng cell wall matrix, exposes more hydrophilic groups, and degrades some insoluble fiber and converts it into the soluble small oligosaccharides (Li et al., 2019). This result is also explained by dextrinization, wherein starch structure is destroyed by high heat (Guha et al., 1997). Amylose (a soluble starch) is split from amylopectin (an insoluble starch), thus increasing the amount of soluble material (Guha et al., 1997). In addition, glycosidic linkages are broken down at high temperature (approximately 80℃), resulting in the release of glucose (a soluble dietary fiber) (Zavareze and Dias, 2011).
WAC is related to the water absorption behavior and particle structures, such as mean particle size, surface area, total pore volume, porosity, and mean pore radius (Yang et al., 2014; Jacobs et al., 2015). Larger particle and rougher surface have larger intraparticulate or interparticulate spaces, which absorb more interstitial water (Zhao et al., 2009; Phat et al. 2015). Furthermore, water accumulated in the pores of plant powders is weakly bonded and can be easily released by centrifugal force, while the water accumulated in the nanopores of plant cell wall matrix or water strongly associated by cell wall polysaccharides through hydrogen bonding can be retained (Jacobs et al., 2015). After ultrafine powderization, porous structure may be destroyed, which leads to decrease in the WAC of RGU powder (Li et al., 2019). The shorter chain length of RGU powder lowers SP. Ultrafine powderization destroys the original plant cell wall matrix of RG destroyed, and decreased the amount of intact macromolecules, both leading to decrease in SP (Tester and Karkalas, 1996).
The increased WSI of RGU powder suggested that ultrafine powderization could increase the solubility of phytochemicals in RGU powder. Ultrafine powderization removes cellulose barrier in plant material, resulting in a smaller particle size, and increases WSI (Lee et al., 2012; Hermansson, 1982). WSI of RGU powder can also be improved by shortening the diffusion time of water soluble molecules in particle (Zhao et al., 2015). In addition, many cracks on the uneven surface of RGU powder may be another important reason for the higher WSI of RGU powder, compared to that of RGC powder (Zhao et al., 2017). Also, the increased WSI of RGU powder is due to the increased surface area, which could expose more polar groups, hydrophilic cellulose, and hemicellulose groups to water (Hong and Zhang, 2005).
3. Effect of ultrafine powderization and hydrogen-rich water extraction on antioxidant activities of red ginseng powders
The antioxidant activities of WGC, RGC, RGU, and RGUH powder were analyzed by TPC, TFC, DPPH, and ABTS. Their results are shown in Table 4.

Total phenolic and total flavonoid contents, and DPPH and ABTS radical scavenging activity of white and red ginseng powders.
WGC, RGC, and RGU powder were extracted with water, but only RGUH powder was extracted with hydrogen-rich water to check the effect of MH dissolved in water. TPCs of WGC and RGC powder were 1.6 and 4.0 mg tannic acid equivalent/g, respectively, and TFCs were 1.2 and 1.6 mg rutin equivalent/g, respectively, as shown in Fig. 6 and Fig. 7. Therefore, the amount of phenolic compounds including flavonoids in the extract of RGC powder was significantly higher than those of WGC powder (p < 0.05). TPCs of RGU and RGUH powder were increased to 6.2 and 7.3 mg tannic acid equivalent/g, respectively, and TFCs to 2.1 and 3.0 mg rutin equivalent/g, respectively.
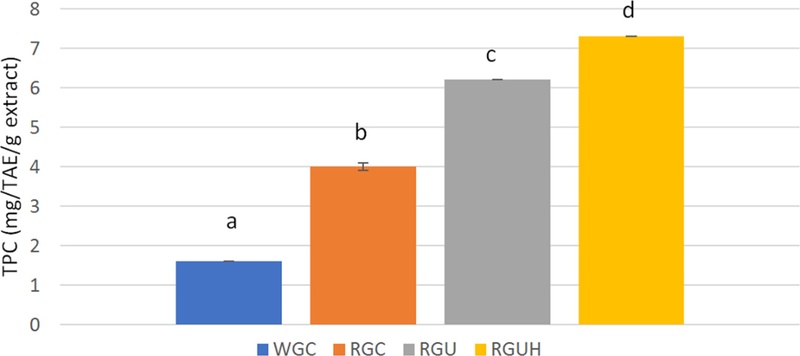
Total phenolic contents of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder; RGUH: hydrogen-rich water extraction of red ginseng ultrafine powder.
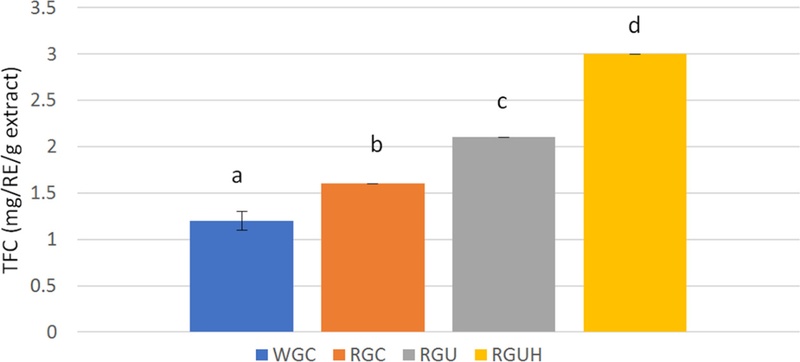
Total flavonoid contents of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder; RGUH: hydrogen-rich water extraction of red ginseng ultrafine powder.
The antioxidant activities of WGC and RGC powder determined by DPPH radical scavenging activity were 51.0% and 86.2%, respectively, and those determined by ABTS radical scavenging activity were 40.7% and 44.9%, respectively. The antioxidant activities of RGU and RGUH powder determined by DPPH radical scavenging activity were increased to 72.7% and 78.6%, respectively, and those determined by ABTS radical scavenging activity to 96.8% and 98.7%, respectively. The antioxidant activities showed significant differences among WGC, RGC, RGU, and RGUH powder (p < 0.05). The antioxidant activities in RGU and RGUH powder were increased markedly with ultrafine powderization and hydrogen-rich water extraction.
DPPH radical scavenging activity has been widely used in determining the antioxidant activities of pure antioxidant compounds and fruit extracts (Shih et al., 2006). DPPH radical scavenging activities in WGC and RGC powder are shown in Table 4 and Fig. 8. In DPPH radical scavenging activity, the WGC and RGC powder scavenged 27.2% and 68.3% of DPPH radicals, respectively. DPPH radical scavenging activity of RGC powder was increased by 1.5 times, compared to that of WGC powder. DPPH radical scavenging activity of RGC powder was significantly (p < 0.05) higher than that of WGC powder.
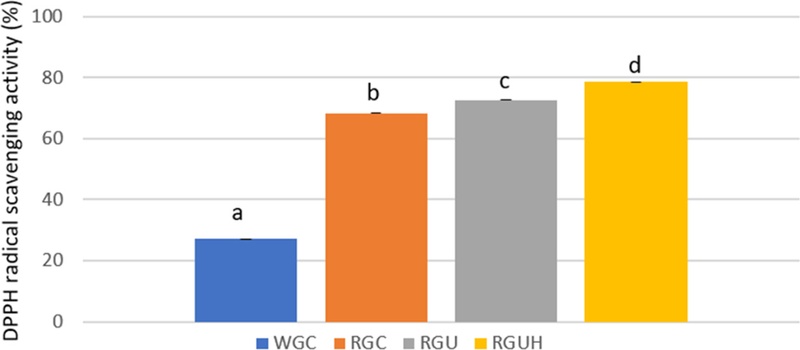
DPPH radical scavenging activity of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder; RGUH: hydrogen-rich water extraction of red ginseng ultrafine powder; DPPH: 2,2-diphenyl-1-picryl-hydrazyl.
ABTS radical scavenging activity has been commonly used to measure the antioxidant activities of various biological specimens (Rufiáan and Morales, 2007). ABTS radical scavenging activities of WGC and RGC powder are shown in Table 4 and Fig. 9. In ABTS radical scavenging activity, WGC and RGC powder scavenged 40.7% and 44.9% of the ABTS radicals, respectively. ABTS radical scavenging activity of RGC powder was increased by 10%, compared to that of WGC powder. ABTS radical scavenging activity of RGC powder was significantly (p < 0.05) higher than that of WGC powder. The antioxidant activity measured by ABTS radical scavenging activity showed the same relationships as DPPH radical scavenging activity did, but antioxidant values were different. This difference in antioxidant activity could be due to the different reaction mechanisms involved. Moreover, it is assumed that the difference in radical scavenging activity is due to different reaction media; ABTS is soluble in both aqueous and organic solvents, allowing for testing in various media, including simulated biological conditions, but DPPH is primarily used in alcoholic solutions (Shalaby and Shanab, 2013).
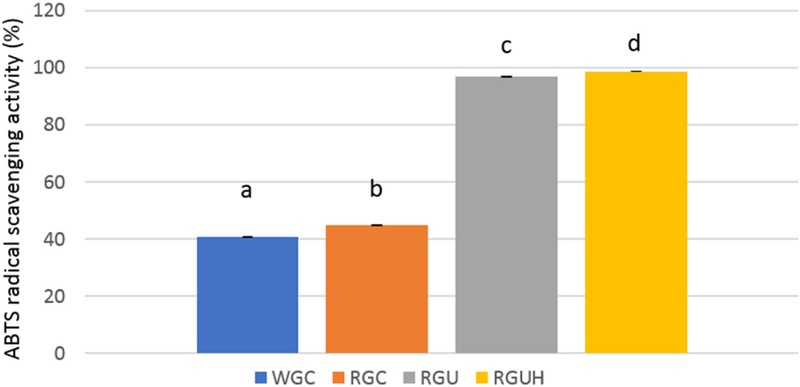
ABTS radical scavenging activity of white and red ginseng powders.Different subscript letters indicate a significantly different (p < 0.05). WGC: White ginseng coarse powder; RGC: Red ginseng coarse powder; RGU: Red ginseng ultrafine powder; RGUH: hydrogen-rich water extraction of red ginseng ultrafine powder; ABTS: 2,2'-azinobis(3-ethylbenzothiazoline-6-sulfonic acid).
More than 10 phenolics in ginseng, including ferulic, gentisic, cinnamic, syringic, and p-hydroxybenzoic acids, have been reported (Dixon and Paiva, 1995). The increased TPC of RGC powder is attributed to the increase of free and conjugated phenolic contents due to the release of phenolic compounds which are bonded with glucosides or amine functionalities during steaming (Dewanto et al., 2002).
TPC and antioxidant activity have a highly positive correlation in many plant species (Oktay et al., 2003). It is known that the antioxidant potentials of phenolic compounds are affected by the quantity and nature of phenolic compounds, such as molecular weight, number of aromatic rings, and nature of hydroxyl group substitution (Li et al., 2008). For these reasons, RGC powder examined in this study demonstrated better antioxidant activity than WGC powder did.
Decrease in particle size results in an increase in macromolecule breakage, specific surface area, exposure of inner pores, and depolymerized cell wall components on particle surfaces (Zhang et al., 2017). Therefore, some phenolic compounds are released or exposed by them (Zhu et al., 2015). Also, this phenomenon is explained by particle size-extraction relationship. Phenolic compounds are easily released, if surface area is increased as particle size is decreased (Cho, 2014) and if ultrafine powderization changes the phenolic compounds to a free form, which is easier to be released from the ginseng cell wall matrix (Kim et al., 2013). When the water extracts reach extraction equilibrium, the microstructural changes affect the diffusion process of phytochemicals, leading to increased extractable macromolecules (Zhang et al., 2017). RGU powder presented higher antioxidant activity than that of RGC powder, which was very likely related to the higher content of phenolic compounds in RGU powder. In this study, the DPPH and ABTS radical scavenging activities of RGU powder showed a similar trend to the results of TPC and TFC, indicating that the DPPH and ABTS radical scavenging activity of RGU powder was related to the amount of phenolic compounds (Rockenbach et al., 2011). It confirms that TPC and TFC were the key factors in antioxidant activities.
When MH was incorporated into water, the TPC and TFC of RGUH powder were increased, compared to those of RGU powder. It can be considered that the increases of TPC and TFC in RGUH powder are due to the protection of phenolic compounds from the oxidative reactions and the liberation of the conjugated and cell wall-bound phenolic compounds (Alwazeer et al., 2023).
Phenolic compounds contain one or more hydroxyl groups (-OH) attached to aromatic ring. These hydroxyl groups are highly susceptible to oxidation reactions. The presence of multiple hydroxyl groups in the phenolic compounds can enhance their susceptibility to oxidation, because each hydroxyl group can act as a potential site of oxidation (Waterhouse and Laurie, 2006). MH has a thermodynamic potential-reducing property that makes it a potent antioxidant. It is involved in the biological electron transfer system at pH 7, with a standard reduction potential of (2H+/H2) that equals -420 mV. Thus, it can shield phenolic compounds from oxidative reactions, making them less susceptible to breakdown (Hu et al., 2021). Therefore, the presence of MH in solvent could potentially protect phenolic compounds from oxidation reactions during the extraction process. This is particularly crucial in cases where the plant materials are subjected to grinding, which can destroy their structure and make them more susceptible to oxygen and oxidation reactions in extraction (Jha and Sit, 2022).
Saturated hydrogen in water easily forms gas bubbles. Hydrogen bubbles burst at solid–liquid interface, leading to the degradation of plant cell wall matrix. Phytochemicals, such as phenolic compounds bound to cell wall, can be liberated after this explosion of bubbles, leading to the dissolution of phenolic compounds in water (Blicharski and Oniszczuk, 2017). In addition, the infusion of MH would have removed any dissolved oxygen. Then it is possible that fewer oxygen radicals (e.g., •, •OH) are produced because there is not enough O2 to react (Duried, 2024). Therefore, MH in solvent can help extract more phytochemicals. For this reason, it can be expected that DPPH and ABTS radical scavenging activities in RGUH powder were improved by the increase of TPC and TFC, compared to those of RGU powder.
In this study, we demonstrate that ultrafine powderization and hydrogen-rich water extraction can be utilized to enhance the antioxidant activity of red ginseng powder by extracting more phenolic compounds. Therefore, both technologies can be applied to other medicinal plants.
References
-
Alwazeer D and Elnasanelkasim MA. (2023). Hydrogen-rich water as a green solvent for the extraction of phytochemicals from agri-food wastes. Sustainable Chemistry and Pharmacy. 33:101035.
[https://doi.org/10.1016/j.scp.2023.101035]

-
Alwazeer D, Elnasanelkasim MA, Çı̇ğdem A, Engin T, Kanmaz H, Hayaloglu AA, Russell G and Hancock JT. (2023). Hydrogen incorporation into solvents can improve the extraction of phenolics, flavonoids, anthocyanins, and antioxidants: A case-study using red beetroot. Industrial Crops and Products. 202:117005.
[https://doi.org/10.1016/j.indcrop.2023.117005]

-
Alwazeer D, Liu FFC, Wu XY and LeBaron WT. (2021). Combating oxidative stress and inflammation in COVID-19 by molecular hydrogen therapy: Mechanisms and perspectives. Oxidative Medicine and Cellular Longevity. 2021:5513868.
[https://doi.org/10.1155/2021/5513868]

-
Ameer K, Shahbaz HM and Kwon J. (2017). Green extraction methods for polyphenols from plant matrices and their byproducts: A review. Comprehensive Reviews in Food Science and Food Safety. 16:295-315.
[https://doi.org/10.1111/1541-4337.12253]

- Andlauer W and Fürst P. (1998). Antioxidative power of phytochemicals with special reference to cereals. Cereal Food World. 43:356-359.
- Azwanida NN. (2015). A review on the extraction methods use in medicinal plants, principle, strength and limitation. Medicinal and Aromatic Plants. 4:412-2167.
-
Blicharski T and Oniszczuk A. (2017). Extraction methods for the isolation of isoflavonoids from plant material. Open Chemistry. 15:34-45.
[https://doi.org/10.1515/chem-2017-0005]

-
Blois MS. (1958). Antioxidant determinations by the use of a stable free radical. Nature. 181:1199-1200.
[https://doi.org/10.1038/1811199a0]

-
Cho YJ. (2014). Antioxidant, angiotensin converting enzyme and xanthin oxidase inhibitory activity of extracts from Saururus chinensis leaves by ultrafine grinding. Food Science and Preservation. 21:75-81.
[https://doi.org/10.11002/kjfp.2014.21.1.75]

-
Choi KT. (2008). Botanical characteristics pharmacological effects and medicinal components of Korean Panax ginseng CA Meyer. Acta Pharmacologica Sinica. 29:1109-18.
[https://doi.org/10.1111/j.1745-7254.2008.00869.x]

-
Dewanto V, Wu X and Liu RH. (2002). Processed sweet corn has higher antioxidant activity. Journal of Agricultural and Food Chemistry. 50:4959-4964.
[https://doi.org/10.1021/jf0255937]

-
Dixon RA and Paiva NL. (1995). Stress-induced phenylpropanoid metabolism. Plant Cell. 7:1085-1097.
[https://doi.org/10.1105/tpc.7.7.1085]

-
Esposito F, Arlotti G, Bonifati AM., Napolitano A, Vitale D and Fogliano V. (2005). Antioxidant activity and dietary fibre in durum wheat bran byproducts. Food Research International. 38:1167-1173.
[https://doi.org/10.1016/j.foodres.2005.05.002]

-
González-Ballesteros-Ballesteros N, Torres MD, Flórez-Fernández N, Diego-González L, Simón-Vázquez R, Rodríguez-Argüelles MC and Domínguez H. (2021). Eco-friendly extraction of Mastocarpus stellatus carrageenan for the synthesis of gold nanoparticles with improved biological activity. International Journal of Biological Macromolecules. 183:1436-1449.
[https://doi.org/10.1016/j.ijbiomac.2021.05.115]

-
Guha M, Ali SZ and Bhattacharya S. (1997). Twin-screw extrusion of rice flour without a die: Effect of barrel temperature and screw speed on extrusion and extrudate characteristics. Journal of Food Engineering. 32:251-267.
[https://doi.org/10.1016/S0260-8774(97)00028-9]

-
Gulsunoglu Z, Karbancioglu-Guler F, Raes K and Kilic-Akyilmaz M. (2019). Soluble and insoluble-bound phenolics and antioxidant activity of various industrial plant wastes. International Journal of Food Properties. 22:1501-1510.
[https://doi.org/10.1080/10942912.2019.1656233]

-
Hermansson AM. (1982). Gel characteristics—structure as related to texture and water binding of blood plasma gels. Journal of Food Science. 47:1965-1972.
[https://doi.org/10.1111/j.1365-2621.1982.tb12924.x]

- Hong J and Zhang SY. (2005). Effect of ultra-fine pulverization by wet processing on particle structure and physical properties of soybean dietary fiber. Journal of South China Agricultural University. 10:90-94
-
Hu H, Li P and Shen W. (2021). Preharvest application of hydrogen-rich water not only affects daylily bud yield but also contributes to the alleviation of bud browning. Scientia Horticulturae. 287:110267.
[https://doi.org/10.1016/j.scienta.2021.110267]

-
Jacobs PJ, Hemdane S, Dornez E, Delcour JA and Courtin CM. (2015). Study of hydration properties of wheat bran as a function of particle size. Food Chemistry. 179:296-304.
[https://doi.org/10.1016/j.foodchem.2015.01.117]

-
Jeon BH, Seong GS, Chun SG, Sung JH and Chang CC. (2005). Antioxidative effects of white ginseng and red ginseng on liver of high fat diet-treated mice. The Korean Society of Ginseng. 29:138-144.
[https://doi.org/10.5142/JGR.2005.29.3.138]

-
Jeong TC, Kim HJ, Park JI, Ha CS, Park JD, Kim SI and Roh JK. (1997). Protective effects of red ginseng saponins against carbon tetrachloride-induced hepatotoxicity in sprague dawley rats. Planta Medica. 63:136-140.
[https://doi.org/10.1055/s-2006-957630]

-
Jha AK and Sit N. (2022). Extraction of bioactive compounds from plant materials using combination of various novel methods: A review. Trends in Food Science and Technology. 119:579-591.
[https://doi.org/10.1016/j.tifs.2021.11.019]

-
Kim HG, Yoo SR, Park HJ, Lee NH, Shin JW, Rekha Sathyanath, Cho JH and Son CG. (2011). Antioxidant effects of Panax ginseng C.A. Meyer in healthy subjects: A randomized, placebo-controlled clinical trial. Food and Chemical Toxicology. 49:2229-2235.
[https://doi.org/10.1016/j.fct.2011.06.020]

-
Kim JS, Kang OJ and Gweon OC. (2013). Comparison of phenolic acids and flavonoids in black garlic at different thermal processing steps. Journal of Functional Foods. 5:80-86.
[https://doi.org/10.1016/j.jff.2012.08.006]

- Kim KY, Shin JK, Lee SW, Yoon SR, Chung HS, Jeong YJ, Chio MS, Lee CM, Moon KD and Kwon JH. (2007). Quality and functional properties of red ginseng prepared with different steaming time and drying method. Korean Journal of Food Science and Technology. 39:494-499.
-
Kim SI, Park JH, Ryu JH, Park JD, Lee YH, Park JH, Kim TH, Kim JM and Baek NI. (1996). Ginsenoside Rg5, a genuine dammarane glycoside from Korean red ginseng. Archives of Pharmacal Research. 19:551-553.
[https://doi.org/10.1007/BF02986026]

-
Kim WY, Kim JM, Han SB, Lee SK, Kim ND, Park MK, Kim CK and Park JH. (2000). Steaming of ginseng at high temperature enhances biological activity. Journal of Natural Product. 63:1702-1704.
[https://doi.org/10.1021/np990152b]

-
Kim YE, Ryu J, Kim JT, Suh S, Hong GP and Ko S. (2018). Physicochemical and in vitro digestion characteristics of size-different red ginseng powders. Food Science and Biotechnology. 27:425-431.
[https://doi.org/10.1007/s10068-017-0254-4]

-
Kirby AR, Ollett AL, Parker R and Smith AC. (1988). An experimental study of screw configuration effects in the twin-screw extrusion-cooking of maize grits. Journal of Food Engineering, 8:247-272.
[https://doi.org/10.1016/0260-8774(88)90016-7]

-
Kitts DD, Wijewickreme AN and Hu C. (2000). Antioxidant properties of a North American ginseng extract. Molecular and Cellular Biochemistry. 203:1-10.
[https://doi.org/10.1023/A:1007078414639]

-
Lee BG, Lee KY, Jorge S, Jorge R, Baek H, Min JH and Kang WS. (2012). Ultrafine powderization using low temperature turbo mill to improve water solubility of red ginseng powder. In Proceedings of the 2012 12th IEEE International Conference on Nanotechnology (IEEE-NANO). IEEE. Birmingham. UK. p. 1-4.
[https://doi.org/10.1109/NANO.2012.6321975]

-
Lee D, Ghafoor K, Moon S, Kim SH, Kim S, Chun H and Park J. (2015). Phenolic compounds and antioxidant properties of high hydrostatic pressure and conventionally treated ginseng(Panax ginseng) products. Quality Assurance and Safety of Crops and Foods. 7:493-500.
[https://doi.org/10.3920/QAS2014.0416]

-
Lee SB, Yoo S, Ganesan P and Kwak HS. (2013). Physicochemical and antioxidative properties of Korean nanopowdered white ginseng. International Journal of Food Science and Technology, 48:2159-2165.
[https://doi.org/10.1111/ijfs.12200]

- Lee SY, Kim YK, Park N I, Kim CS, Lee CY and Park SU. (2010). Chemical constituents and biological activities of the berry of Panax ginseng. Journal of Medicinal Plants Research. 4:349-353.
-
Li B, Yang W, Nie Y, Kang F, Goff HD and Cui SW. (2019). Effect of steam explosion on dietary fiber, polysaccharide, protein and physicochemical properties of okara. Food Hydrocolloids. 94:48-56.
[https://doi.org/10.1016/j.foodhyd.2019.02.042]

-
Li HB, Wong CC, Cheng KW and Chen F. (2008). Antioxidant activities in vitro and total phenolic contents in methanol extracts from medicinal plants. LWT - Food Science and Technology. 41:385-390.
[https://doi.org/10.1016/j.lwt.2007.03.011]

- Matsuura Y, Zheng Y, Takaku T, Kameda K and Okuda H. (1994). Isolation and physiological activities of a new amino acid derivative from Korean red ginseng. Journal of Ginseng Research. 18:204-2011.
-
Moreno MI, Isla MI, Sampietro AR and Vattuone MA. (2000). Comparison of the free radical-scavenging activity of propolis from several regions of Argentina. Journal of Ethnopharmacology. 71:109-114.
[https://doi.org/10.1016/S0378-8741(99)00189-0]

-
Nam KY. (2005). The comparative understanding between red ginseng and white ginseng. Journal of Ginseng Research. 29:1-18.
[https://doi.org/10.5142/JGR.2005.29.1.001]

-
Oktay M, Culcin I and Kufrevioglu OI. (2003). Determination of in vitro antioxidant activity of ennel(Foenniculum vulgare) seed extracts. LWT - Food Science and Technology. 36:263-271.
[https://doi.org/10.1016/S0023-6438(02)00226-8]

-
Park MJ, Kim MK, In JG and Yang DC. (2006). Molecular identification of Korean ginseng by amplification refractory mutation system-PCR. Food Research International. 39:568-574.
[https://doi.org/10.1016/j.foodres.2005.11.004]

-
Phat C, Li H, Lee DU, Moon BK, Yoo YB and Lee C. (2015). Characterization of Hericium erinaceum powders prepared by conventional roll milling and jet milling. Journal of Food Engineering. 145:19-24.
[https://doi.org/10.1016/j.jfoodeng.2014.08.001]

-
Rao P and Rathod V. (2019). Valorization of food and agricultural waste: A step towards greener future. The Chemical Record. 19:1858-1871.
[https://doi.org/10.1002/tcr.201800094]

-
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M and Rice-Evans C. (1999). Antioxidant activity applying an improved ABTS radical cation decolourization assay. Free Radical Biology and Medicine. 26:1231-1237.
[https://doi.org/10.1016/S0891-5849(98)00315-3]

-
Régnier T and Macheix JJ. (1996). Changes in wall-bound phenolic acids, phenylalanine and tyrosine ammonia-lyases, and peroxidases in developing durum wheat grains(Triticum turgidum L. var. durum). Journal of Agricultural and Food Chemistry. 44:1727-1730.
[https://doi.org/10.1021/jf950607c]

-
Rockenbach II, Gonzaga LV, Rizelio VM, Gonçalves AESS, Genovese MI and Fett R. (2011). Phenolic compounds and antioxidant activity of seed and skin extracts of red grape(Vitis vinifera and Vitis labrusca) pomace from Brazilian winemaking. Food Research International. 44:897-901.
[https://doi.org/10.1016/j.foodres.2011.01.049]

-
Rosa-Sibakov N, Sibakov J, Lahtinen P and Poutanen K. (2015). Wet grinding and microfluidization of wheat bran preparations: Improvement of dispersion stability by structural disintegration. Journal of Cereal Science. 64:1-10.
[https://doi.org/10.1016/j.jcs.2015.04.002]

-
Rufián-Henares JA and Morales FJ. (2007). Functional properties of melanoidins: In vitro antioxidant, antimicrobial, and antihypertensive activities. Food Research International. 40:995-1002.
[https://doi.org/10.1016/j.foodres.2007.05.002]

-
Seo SJ, Cho JY, Jeong YH and Choi YS. (2013). Effect of Korean red ginseng extract on liver damage induced by short-term and long-term ethanol treatment in rats. Journal of Ginseng Research. 37:194-200.
[https://doi.org/10.5142/jgr.2013.37.194]

- Shalaby EA and Shanab SM. (2013). Comparison of DPPH and ABTS assays for determining antioxidant potential of water and methanol extracts of Spirulina platensis. Indian Journal of Geo-Marine Sciences. 42:556-564.
-
Shih PW, Lai PL and Jen HWK. (2006). Antioxidant activities of aqueous extracts of selected plants. Food Chemistry. 99:775-783.
[https://doi.org/10.1016/j.foodchem.2005.07.058]

- Tester RF and Karkalas JO. (1996). Swelling and gelatinization of oat starches. Cereal Chemistry. 73:271-277.
- Tiwari BK, Brunton N and Brennan CS. (2015). Handbook of plant food phytochemicals. Wiley Online Library. Chichester. England.
-
Waterhouse AL and Laurie VF. (2006). Oxidation of wine phenolics: A critical evaluation and hypotheses. American Journal of Enology and Viticulture. 57:306-313.
[https://doi.org/10.5344/ajev.2006.57.3.306]

-
Williamson EM, Liu X and Izzo AA. (2020). Trends in use, pharmacology, and clinical applications of emerging herbal nutraceuticals. Britsh Journal of Parmacology. 177:1227-1240.
[https://doi.org/10.1111/bph.14943]

- Yang SJ, Woo KS, Yoo JS, Kang TS, Noh YH, Lee JS and Jeong HS. (2006). Change of Korean ginseng components with high temperature and pressure treatment. Korean Journal of Food Science and Technology. 38:521-525.
-
Yang Y, Ji G, Xiao W and Han L. (2014). Changes to the physicochemical characteristics of wheat straw by mechanical ultrafine grinding. Cellulose. 21:3257-3268.
[https://doi.org/10.1007/s10570-014-0381-5]

-
Yu L, Haley S, Perret J and Harris M. (2002a). Antioxidant properties of hard winter wheat extracts. Food Chemistry. 78:457-461.
[https://doi.org/10.1016/S0308-8146(02)00156-5]

-
Yu L, Haley S, Perret J, Harris M, Wilson J and Qian M. (2002b). Freeradical scavenging properties of wheat extracts. Journal of Agricultural and Food Chemistry. 50:1619-1624.
[https://doi.org/10.1021/jf010964p]

- Yun TK and Lee YS. (1994a). Anticarcinogenic effect of ginseng powders depending on the types and ages using Yun's anticarcinogenicity test(I). Journal of Ginseng Research, 18:89-94.
- Yun TK and Lee YS. (1994b). Anticarcinogenic effect of ginseng powders depending on the types and ages using Yun's anticarcinogenicity test(II). Journal of Ginseng Research, 18:160-164.
- Yun TK, Lee YS, Kwon HY and Choi KJ. (1996). Saponin contents and anticarcinogenic effects of ginseng depending on types and ages in mice. Acta Pharmacologica Sinica. 17:293-298.
- Yun TK, Yun YS and Han IW. (1983). Anticarcinogenic effect of long-term oral administration of red ginseng on newborn mice exposed to various chemical carcinogens. Cancer Detection and Prevention. 6:515-525.
-
Zavareze EDR and Dias ARG. (2011). Impact of heat-moisture treatment and annealing in starches: A review. Carbohydrate Polymers. 83:317-328.
[https://doi.org/10.1016/j.carbpol.2010.08.064]

-
Zhang Y, Xiao W, Ji G, Gao C, Chen X, Cao Y and Han L. (2017). Effects of multiscale-mechanical grinding process on physicochemical properties of black tea particles and their water extracts. Food and Bioproducts Processing. 105:171-178.
[https://doi.org/10.1016/j.fbp.2017.05.002]

-
Zhao X, Yang Z, Gai G and Yang Y. (2009). Effect of superfine grinding on properties of ginger powder. Journal of Food Engineering. 91:217-222.
[https://doi.org/10.1016/j.jfoodeng.2008.08.024]

-
Zhao X, Zhu H, Zhang G and Tang W. (2015). Effect of superfine grinding on the physicochemical properties and antioxidant activity of red grape pomace powders. Powder Technology. 286:838-844.
[https://doi.org/10.1016/j.powtec.2015.09.025]

-
Zhao Y, Wu X, Wang Y, Jing R and Yue F. (2017). Comparing physicochemical properties of hawthorn superfine and fine powders. Journal of Food Processing and Preservation, 41:e12834.
[https://doi.org/10.1111/jfpp.12834]

-
Zhu FM, Du B and Xu BJ. (2015). Superfine grinding improves functional properties and antioxidant capacities of bran dietary fibre from Qingke(hull-less barley) grown in Qinghai–Tibet Plateau, China. Journal of Cereal Science. 65:43-47.
[https://doi.org/10.1016/j.jcs.2015.06.006]

