Protective Effects of Agastache rugose Against Cyclophosphamide-Induced Cognitive Impairment in Mice: Experimental Validation and Compound-Target Prediction
Correspondence to: #Huiyeong Jeong and Youngmee Kim are contributed equally to this paper.
This is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
The chemotherapeutic and immunosuppressive agent cyclophosphamide (CYP), which is widely used in cancer treatment, can cause oxidative damage and inflammation in the brain, resulting in behavioral abnormalities, including cognitive impairment. Agastache rugosa (AR) has been traditionally used to treat digestive disorders and it has several physiological activities.
We investigated the neuroprotective effects of an AR ethanol extract against CYP-induced neurodegeneration and cognitive dysfunction. Free radical scavenging assays confirmed the antioxidant activity of the AR extract. C57BL/6 mice were orally administered an AR extract (100 or 200 mg/kg) for seven days, followed by an intraperitoneal injection of CYP (50 mg/kg). Mice exhibited memory impairment in a contextual fear conditioning test 24 h after CYP treatment. However, AR pretreatment notably improved behavioral deficits. Histological analysis demonstrated that CYP administration reduced the immunoreactivity of Ki67 (a proliferative cell marker) and doublecortin (an immature progenitor cell marker) in the dentate gyrus of the hippocampus, whereas AR pretreatment considerably normalized the levels of these markers. Subsequently, network pharmacology was used to investigate the potential genes involved in the ameliorative action of AR on CYP-induced neurodegeneration and memory impairment, which suggested BCL2, JUN, and MAPK3 as key candidates.
These findings demonstrate that AR pretreatment protects against CYP-induced neurodegeneration and cognitive impairment. In addition, network pharmacology analysis suggests that AR compounds may provide neuroprotection against CYP-induced neuroinflammation by modulating key target genes such as BCL2, JUN, and MAPK3.
Keywords:
Agastache rugosa, Cyclophosphamide, Cognitive Impairment, Antioxidant Activity, Network Pharmacology AnalysisINTRODUCTION
Cyclophosphamide (CYP) has been extensively used as an anticancer and immunosuppressive agent (de Jonge et al., 2005). Although the chemotherapeutic agent was initially thought not to cross the blood-brain barrier when administered at standard clinically relevant doses, 17 - 75% of patients, who received the chemotherapy, have reported several adverse effects related to learning and memory such as concentration difficulties and cognitive impairment (Silberfarb, 1983; Hodgson et al., 2013; Lange et al., 2014; Matsos and Johnston, 2019). Furthermore, the extent of cognitive dysfunction was proportional to the dose of chemotherapy (Ahles and Saykin, 2001). Several molecular mechanisms underlying the CYP-induced deficits in learning and memory function have been studied, which include oxidative stress, inflammation, neuronal cell death, and interference with the function of neurotransmitters and neurotrophic factors (Bagnall-Moreau et al., 2019).
Agastache rugosa (AR), also known as Korean mint, is a member of the Lamiaceae family (Rinik et al., 2024), and it is widely distributed across East Asia, including Korea, China, Japan, and Russia. AR, a major component of the traditional formula Gwakhyangjeonggi-san, has been historically used for digestive disorders, including diarrhea, abdominal discomfort, and vomiting (Ko et al., 2013). However, recent accumulating pharmacological evidence suggests that AR may broaden the therapeutic potential beyond classical indications by its antioxidant, anti-inflammatory, anti-aging, anti-bacterial, and anti-tumor activities. These effects are largely attributed to its bioactive constituents, including flavonoids (e.g., acacetin, tilianin), phenylpropanoids (e.g., rosmarinic acid), lignans, and terpenoids (Nam et al., 2020; Song et al., 2024). In this study, we aimed to broaden the indications of AR treatment by investigating its ameliorative effects in a mouse model of neurodegeneration and memory impairment induced by CYP administration. Moreover, we examined the possible target genes under the protective effects of AR against CYP-induced neurotoxicity by using network pharmacology analysis, which has been widely applied to explain the multi-component, multi-target, and multi-pathway behavior of medicinal herbs (Song et al., 2024). To this end, we performed the analysis using encephalitis (a clinical diagnosis broadly regarded as reflecting neuroinflammatory conditions) as a keyword, allowing us to identify the predicted target genes potentially related to neuroinflammatory processes.
MATERIALS AND METHODS
1. 70% ethanol extract of AR
The AR extract was prepared as previously published (Kang et al., 2024). Briefly, the whole shoot part of AR was obtained from Jeju Island (Seongsan-eup, Seogwipo-si, Republic of Korea [33°27′47.5″ N, 126°54′48.9″ E, altitude 17 m]). Total 100 g of the dried AR plant was included in 1 L of 20% EtOH (v/v) solution, and refluxed for 2 h at 30°C. The extract was filtered, concentrated with a rotary air evaporator, and lyophilized using a freeze dryer. The final extract was stored at −80°C until use.
2. DPPH and ABTS radical scavenging activity
1,1-diphenyl-2-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) free radical scavenging activities for measurement of antioxidant capacities were determined according to report (n = 5 per each group) (Seo et al., 2025). DPPH solution dissolved in methanol and 100 μL AR extract (0, 5, 10, 20, 100, and 500 mg/mL) reacted at 37°C in an incubator for 30 min. The absorbance of 100 μL DPPH radical was measured at 517 nm. For the ABTS assay, the radical solution reacted with phosphate was adjusted to a final absorbance of 0.7 ± 0.02 at 734 nm. Each scavenging rate of DPPH and ABTS of the AR extract was calculated according to the following formula:
Scavenging rate (%) = [(Abfree − Absample)/Abfree] × 100
3. Animals
Seven-week-old male C57BL/6 mice (18 ± 2 g) were obtained from Dooyeol Biotech (Seocho-gu, Seoul, Korea). The mice were housed in standard mouse cages at a temperature of 23 ± 2°C, humidity of 50 ± 10%, air ventilation frequency of 10 to 20 times/h, and light intensity of 105 to 300 Lux from 08:00 to 20:00, with food and water provided randomly. All mice were housed in accordance with the guidelines set forth in the Animal Welfare Regulation, and the experimental procedures were approved by the Institutional Animal Care and Use Committee of Chonnam National University (CNU IACUC-YB-2024-112) and Korea Institute of Oriental Medicine (20-061, 01 September 2020).
4. CYP and AR extract administration
Following an acclimatization period, the mice were divided into four groups (n = 6 per each group). The first group served as control (control group), and the second group was administered only CYP (CYP group). Group 3 was administered CYP and AR extract at a dose of 100 mg/kg (CYP+AR100), whereas group 4 received the same treatment but with a higher dose of AR extract of 200 mg/kg (CYP+AR200). The AR treatment groups were administered 100 or 200 mg/kg/day of the extract for 7 days. Mice in groups 2, 3, and 4 were intraperitoneally injected with CYP (50 mg/kg) on day 7, whereas group 1 was administered phosphate-buffered saline (PBS) instead of CYP.
5. Contextual fear conditioning test
Contextual fear conditioning paradigm was conducted by using a TruScan Photobeam Activity Monitoring System (n = 6 per each group) (Coulbourn Instruments, Whitehall, PA, USA). Each mouse was placed in the conditioning chamber (26 × 26 × 40 cm) with 23 stainless steel grids on the floor, and allowed to explore the space for 2 min before an electronic impact. Subsequently, the subjected mouse received a single shock (0.5 mA for 2 s) and was permitted to remain in the conditioning chamber for 30 s. 24 hours following the training, the mice were returned to the conditioning chamber for 2 min to assess the percentage of the complete absence of movement behavior (defined as freezing) except for respiration. The duration of the freezing behavior was recorded when the mouse exhibited sustained freezing for a minimum of 2 seconds.
6. Immunohistochemical analysis
Mice were euthanized by the combination of alfaxalone and xylazine, and the brains were removed immediately, and fixed in 4% paraformaldehyde for paraffin embedding (n = 3 per each group). The immunohistochemical analysis was conducted in accordance with previously described protocols in the literature (Lee et al., 2022). Paraffin sections (thickness, 4 μm) were cut with a microtome, and deparaffinized using routine protocols, and tissue sections were treated with primary antibodies. The primary antibodies used were rabbit anti-Ki67 (1:100; #DRM004, OriGene Technologies Inc., Beijing, China) and rabbit anti-DCX (1:1,000; #4604, Cell Signaling Technology, Beverly, MA, USA), diluted in antibody dilution buffer (Invitrogen, Carlsbad, CA, USA). Subsequently, the sections were washed with PBS and incubated with biotinylated goat anti-rabbit IgG (Vector ABC Elite Kit; Vector Laboratories, Burlingame, CA, USA). The specific binding was detected with the Vector ABC Elite Kit (Vector Laboratories) employing an avidin-biotin-peroxidase complex, and stained utilizing a diaminobenzidine (DAB; Vector Laboratories) substrate. The sections were then counterstained with hematoxylin prior to mounting, and the DAB-positive cells were analyzed using a microscope platform (BX53 apparatus; Olympus, Tokyo, Japan).
7. Network pharmacology analysis
The sample used in this study has been mentioned in the previous reports, and the components used in the network analysis were only detected by liquid chromatography and gas chromatography (Nam et al., 2020; Song et al., 2024). These components were selected using the in silico integrative Absorption, Distribution, Metabolism, and Excretion (ADME) model, based on which the components were screened for Oral Bioavailability (Impey et al., 1999) ≥ 30.0% and -0.3 < Blood-Brain Barrier (BBB) according to Traditional Chinese Medicine System Pharmacology Database (TCMSP) (https://www.tcmspe.com/index.php). Drug Likeness (Sekeres et al., 2021) values were ‘yes’ according to SwissADME databases (http://www.swissadme.ch/). The chemical information of all components was confirmed at PubChem (https://pubchem.ncbi.nlm.nih.gov/) and ChemSpider (https://www.chemspider.com/) databases.
Information on the proteins related to active components in AR was collected from the Search Tool for Interactions of Chemicals and Proteins (STITCH) database (http://stitch.embl.de/, ver. 5.0) with ‘Homo sapiens’ and SwissTargetPrediction (http://www.swisstargetprediction.ch/) databases (Park et al., 2022). Information on genes was verified in the UniProt database (https://www.uniprot.org/). The Venn diagram was drawn using Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/).
Potential target genes were defined as proteins that overlapped with the protein-derived active components and proteins linked to encephalitis. Information on encephalitis-derived proteins was searched for the GeneCards: The Human Gene Database (https://www.genecards.org/, version 5.19). The PPI network was built after searching the STRING database (https://string-db.org/, version 12) with ‘Homo sapiens’, and the number of first shell interactors did not exceed 50 with a confidence score ≥ 0.400 (medium confidence) (Park et al., 2022). Topology analysis including degree, closeness, and betweenness centrality of the PPI network was conducted using Cytoscape version 3.10.1 (https://cytoscape.org/).
Signaling pathways were analyzed using Database for Annotation, Visualization and Integrated Discovery (DAVID) (https://david.ncifcrf.gov/, updated January 12, 2024 version) and Kyoto Encyclopedia of Genes and Genomes (KEGG) (https://www.genome.jp/kegg/) with p < 0.05. The network was visualized using Cytoscape version 3.10.1 (Cytoscape, Boston, MA, USA).
8. Statistical analysis
The analysis parameters are presented as mean ± standard error (SE). Statistical significance was identified by one-way analysis of variance, followed by Holm-Sidak post hoc test using GraphPad Prism 10 (GraphPad software, San Diego, CA, USA). Statistical significance was set at p < 0.05.
RESULTS
1. The antioxidative activities of AR extract in DPPH- and ABTS-scavenging assays
To determine the antioxidant activity of AR extract, we measured free radical scavenging rates in DPPH and ABTS assays at different concentrations of the extracts (0, 5, 10, 20, 100, and 500 mg/mL; Fig. 1). AR extracts at concentrations ≥ 20 mg/mL exerted significantly higher antioxidative activities in DPPH (p = 0.610 and p < 0.001 for the other comparisons) and ABTS (p = 0.009 and p < 0.001 for remaining comparisons) assays compared with AR 5 mg/mL-treated group. At concentrations below 100 mg/mL, the AR extract exhibited a steeply increasing antioxidant effect in a dose-dependent manner, whereas the slope of scavenging rates became gentler above 100 mg/mL.
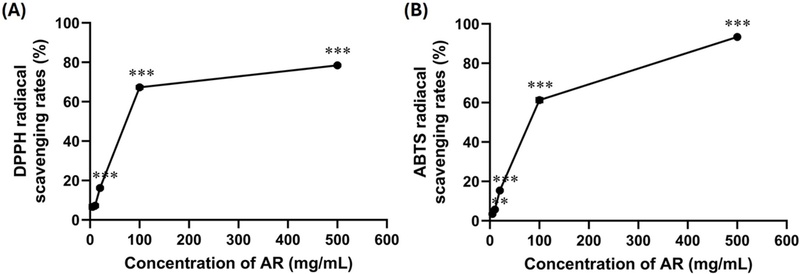
The percentage of DPPH (A) and ABTS (B) radical scavenging activity showed the antioxidant benefits of AR.All data are presented as mean ± standard error (SE). **p < 0.01 and ***p < 0.001 indicates a significant difference compared to AR 5 mg/mL-treated group. DPPH, 1,1-diphenyl-2-picrylhydrazyl; ABTS, 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid); AR, Agastache rugosa extract.
2. Protective effects of AR extract in the CYP-induced cognitive impairment
We evaluated memory and cognitive performance by measuring the freezing time during the contextual fear conditioning test (Fig. 2). There were no differences between groups in the training phase, but in the testing phase, the CYP group showed a significantly lower freezing time than that of the control group (p = 0.015). However, AR pretreatment before CYP administration tended to increase the freezing time, although the significance was observed in only CYP+AR200 group (p = 0.040).
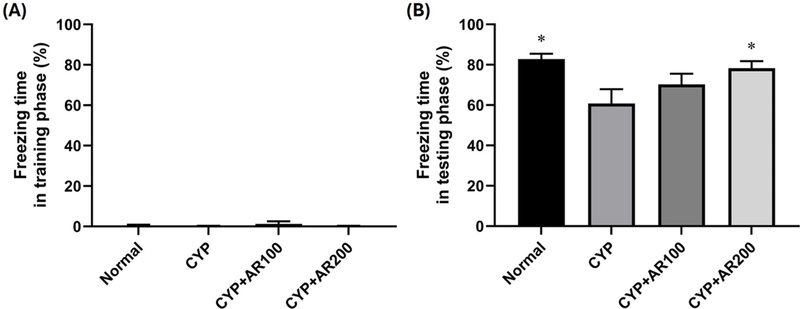
Freezing time (%) during training (A) and testing (B) phases of the contextual fear conditioning test.All data are presented as mean ± standard error (SE). *p < 0.05 indicates a significant difference compared to CYP group. CYP, cyclophosphamide treated group; CYP+AR100, cyclophosphamide and Agastache rugosa extract at a dose of 100 mg/kg treated group; CYP+AR200, cyclophosphamide and Agastache rugosa extract at a dose of 200 mg/kg treated group.
3. Ameliorative effect of AR extract against CYP-induced suppression of hippocampal neurogenesis
Proliferating progenitor cells and immature neurons in the dentate gyrus (DG) of adult mouse hippocampus were quantified based on the number of Ki67- and DCX-positive cells, respectively (Fig. 3). Representative microscopic images are presented in Fig. 3A. CYP administration significantly decreased the positive cells of Ki67- (Fig. 3B) and DCX-positive cells (Fig. 3C) compared with the control group (p = 0.016 and p = 0.033, respectively). However, pretreatment with AR at 100 and 200 mg/kg ameliorated the reduction of Ki67-positive cells (both p = 0.016), and 100 mg/kg of AR significantly increased the expression of DCX-positive cells (p = 0.033).
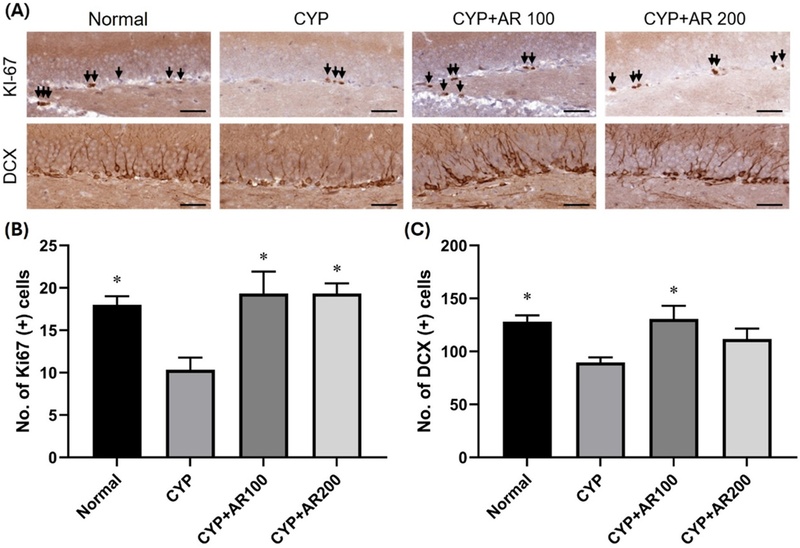
Pretreatment of AR extract improved the decrease in the number of Ki67- and DCX-positive cells in the subgranular zone of the hippocampus after CYP treatment.(A) Representative microscopic images of Ki67- (arrows) and DCX-positive cells in the hippocampal DG. Quantitative analysis of Ki67-positive cells (B) and DCX-positive cells (C). Scale bar is 100 μm. All data were expressed as mean ± standard error (SE). *p < 0.05 indicates a significant difference compared to CYP group. CYP, cyclophosphamide treated group; CYP+AR100, cyclophosphamide and Agastache rugosa extract at a dose of 100 mg/kg treated group; CYP+AR200, cyclophosphamide and Agastache rugosa extract at a dose of 200 mg/kg treated group. DCX: Doublecortin.
4. Active small molecules and target genes
The literature search was conducted for 11 active components identified through liquid and gas chromatographic analyses in a 70% ethanol extract of AR (Nam et al., 2020; Song et al., 2024). These components were rosmarinic acid, tilianin, acacetin 7-O-(6"-O-malonyl)-beta-D-glucopyranoside, isoagastachoside, acacetin 7-O-(2"-O-acetyl-6"-malony)-beta-D-glucopyranoside, acacetin, limonene, menthone, estragole, pulegone, and methyl eugenol. Rosmarinic acid and tilianin did not pass the ADME criteria according to public databases. Acacetin 7-O-(6"-O-malonyl)-beta-D-glucopyranoside, isoagastachoside, and acacetin 7-O-(2"-O-acetyl-6"-malony)-beta-D-glucopyranoside were excluded because of lack of related ADME data from public databases. Finally, six components including acacetin, limonene, menthone, estragole, pulegone, and methyl eugenol were selected. Their OB was ≥ 30 % and -0.3 < BBB according to TCMSP. DL values were ‘yes’ according to SwissADME databases. Structurally, these components were classified as one flavonoid (acacetin) and five monoterpenes (limonene, menthone, estragole, pulegone, and methyl eugenol). The monoterpenes had higher OB, DL, and BBB values than the flavonoids (Table 1).
5. Potential target genes and PPI
Based on STITCH and SwissTargetPrediction databases, 659 genes associated with the six active components of AR were identified, and 2,290 encephalitis-related proteins from the GeneCards database were retrieved. The overlap between the two datasets resulted in 161 common proteins (Fig. 4A). The network between 161 potential target proteins and six active components is shown in Fig. 4B. This network consisted of 167 nodes and 285 edges, of which ACHE, KDR, and PARP1 were the core proteins associated with five active components. ALOX5, CYP1A2, HMOX1, MAOB, MPO, NOS2, and SRD5A1 were related to four active components. CCR5, which had a high relevance score with encephalitis in the GeneCards database, had links to limonene and pulegone.
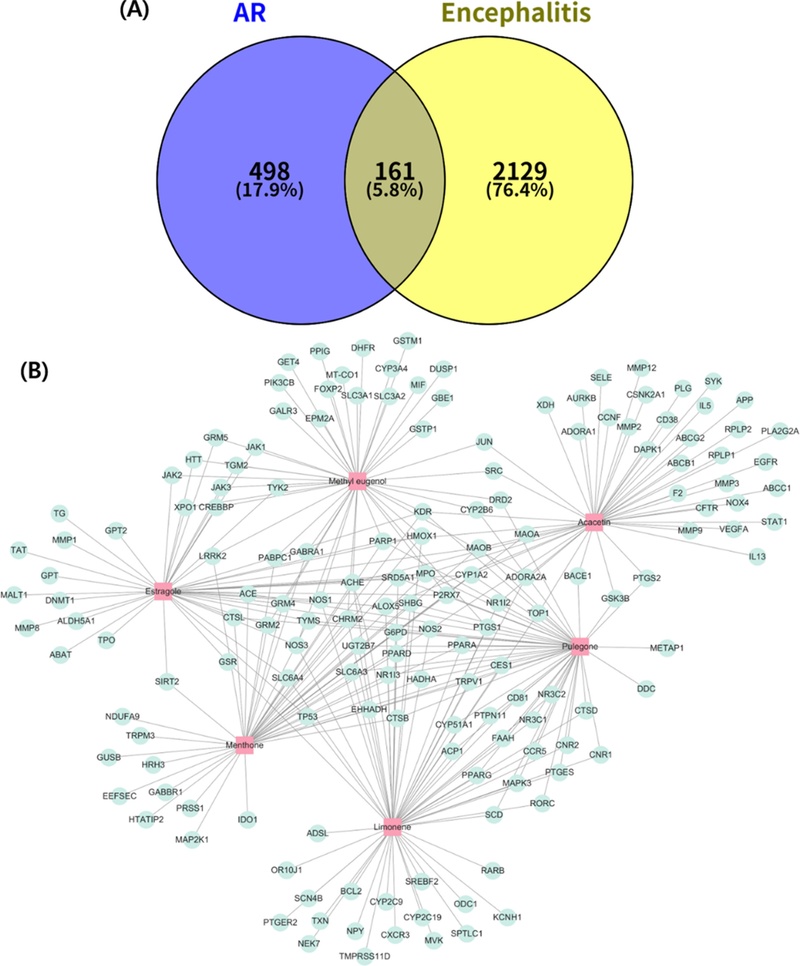
Network of six active components–161 potential target proteins related to encephalitis.(A) Venn diagram showing the intersection of 659 proteins associated with AR and 2,290 proteins involved in encephalitis. (B) The network comprised 167 nodes and 285 edges. Pink rectangles are active components, and light blue ovals represent potential target proteins. AR, Agastache rugosa extract.
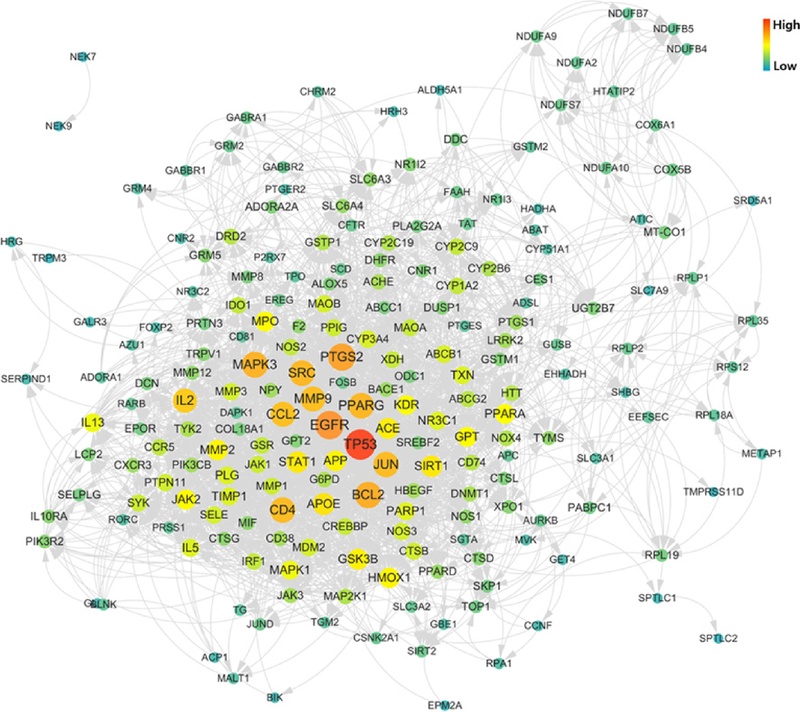
We conducted the PPI analysis for analyzing the interaction of 161 target proteins using the frequently used STRING database (Wei et al., 2021) and visualized the PPI network using Cytoscape 3.10.1. The topological characteristics are calculated by the “network analyzer” function of this software (Chen et al., 2021; Gao et al., 2021). The genes with a high confidence score were TP53, BCL2, CCL2, CD4, EGFR, IL2, JUN, MAPK3, MMP9, PPARG, PTGS2, and SRC (Fig. 5). A high confidence score indicates strong evidence for functional or physical relationship between target proteins. The top 10 proteins of edge count were TP53, EGFR, PTGS2, BCL2, MAPK3, SRC, PPARG, JUN, CD4, and CCL2. The top 10 proteins of betweenness centrality were EGFR, MAPK3, IL2, PPARG, GPT, MMP9, JUN, BCL2, RPL19, and DRD2. The betweenness centrality indicates the frequency with which the shortest paths between any pair of nodes pass through that node; a larger value in the network indicates that the node has greater importance (Lee et al., 2020).
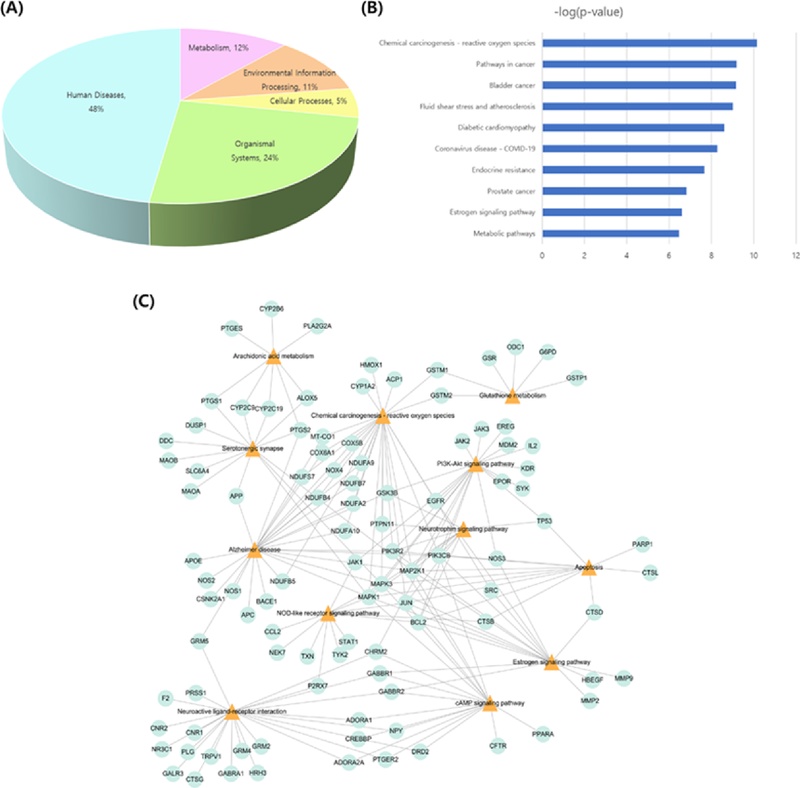
6. Pathway analysis related to encephalitis
The signaling pathways were analyzed using the DAVID and KEGG databases with the p-value (p < 0.05) correction algorithm (Fig. 6) (Xu et al., 2022). A total of 128 pathways were searched in the DAVID database, 48% of which were pathways classified into the category of human diseases (Fig. 6A), and the top 10 pathways are shown in Fig. 6B. The 12 pathways related to encephalitis were serotonergic synapse, estrogen signaling pathway, neuroactive ligand-receptor interaction, chemical carcinogenesis-reactive oxygen species cAMP signaling pathway, arachidonic acid metabolism, apoptosis, PI3K-Akt signaling pathway, NOD-like receptor signaling pathway, neurotrophin signaling pathway, Alzheimer’s disease, and glutathione metabolism. A total of 86 potential target proteins were involved in the encephalitis-related pathways (Fig. 6C). The six proteins BCL2, JUN, MAP2K1, MAPK1, MAPK3, and PIK3CB were associated with more than five pathways.
DISCUSSION
In this study, we demonstrated the neuroprotective effects of AR extract in a mouse model of CYP-induced neurodegeneration. We confirmed the anti-oxidant activity of AR extract. Additionally, pretreatment of AR extract significantly improved the cognitive impairment in contextual fear conditioning test and restored the reduced immunoreactivities of DCX and Ki67 in the hippocampus of CYP-treated mouse. Furthermore, by analyzing active ingredients of AR extract based on the published literature and conducting a network pharmacology analysis of AR-related target genes in CYP-induced neuroinflammation, we predicted several potential target genes that may be involved in the protective effects of AR extract against CYP-induced neurodegeneration.
Although chemotherapy is known to cause cognitive impairment in cancer patients, the underlying mechanisms remain poorly understood. Previous studies have shown a link between chemotherapy-induced hippocampal-dependent memory deficits and impairment in hippocampal neurogenesis (Sekeres et al., 2021). Similarly, in the present study, we identified cognitive impairment and decrease of hippocampal neurogenesis in CYP-administered mice. Among several mechanisms underlying CYP-induced neurogenesis impairment, neuroinflammation and oxidative stress appear to play critical roles. Intraperitoneal injection of CYP induces inflammatory responses in the central nervous system, leading to the activation of glial cells (Hirshman et al., 2020). Activated glial cells secrete inflammatory cytokines, which could inhibit the proliferation and differentiation of neural progenitor cells in the hippocampal DG and damage the neurogenic microenvironment (Monje et al., 2003; Sato, 2015). In parallel, CYP metabolism generates reactive oxygen species and toxic aldehyde byproducts, which induce oxidative stress in neural stem cells, subsequently leading to the decline in neurogenesis (Hussein et al., 2024). CYP-induced impairment in neurogenesis and synaptic plasticity, likely driven by neuroinflammation and oxidative stress, could ultimately be associated with cognitive impairment (Ibrahim et al., 2024). Therefore, targeting neuroinflammation and oxidative stress may offer a promising strategy to ameliorate the chemotherapy-induced cognitive deficits.
AR has been traditionally used to treat digestive disorders, including diarrhea, abdominal discomfort, and vomiting (Ko et al., 2013). Several studies have shown that AR extract and its compounds exhibit various therapeutic effects including anti-oxidant (Desta et al., 2016), anti-microbial (Shin, 2004), anti-inflammatory (Zielińska and Matkowski, 2014), and cardiovascular effects (Cao et al., 2017). To explore the target genes potentially involved in the therapeutic effects of AR against CYP-induced neuroinflammation, we conducted a network pharmacology analysis based on AR compounds identified in our previous study (Nam et al., 2020; Song et al., 2024). 11 constituents of AR were classified as five flavonoids (acacetin, tilianin, acacetin 7-O-(6"-O-malonyl)-beta-D-glucopyranoside, isoagastachoside, acacetin 7-O-(2"-O-acetyl-6"-malony)-beta-D-glucopyranoside), one phenylpropanoid (rosmarinic acid) and five monoterpenes (limonene, menthone, estragole, pulegone, and methyl eugenol). Among the 11 components, five were excluded due to lack of information in the databases or failure to meet the ADME criteria. Subsequent network analysis showed that five small molecules including limonene, menthone, estragole, pulegone, and methyl eugenol had high values of OB, DL, and BBB permeability. These five compounds exhibit significant potential in improving cognitive function through various mechanisms including anti-inflammation, anti-oxidation and neuroprotection (de Sousa et al., 2024; Eddin et al., 2021; Gogoi et al., 2020; Park et al., 2022; Roy et al., 2018; Wang and Heinbockel, 2018). These characteristics collectively indicate that the AR compounds could be promising candidates for the treatment of neurodegenerative diseases.
Next, to identify potential genes associated with the therapeutic effects of AR compounds in CYP-induced neuroinflammation, we performed a target prediction analysis followed by PPI network and functional enrichment analyses. The 659 proteins associated with AR and the 2,290 encephalitis-related proteins formed an intersection of 161 proteins. Analysis of the interactions between 161 proteins revealed that TP53, BCL2, CCL2, CD4, EGFR, IL2, JUN, MAPK3, MMP9, PPARG, PTGS2, and SRC were key nodes exerting significant influence within the PPI network. Among the proteins, BCL2, EGFR, JUN, MAPK3, and PPARG were identified as overlapping proteins with both the top 10 proteins ranked by edge count and the top 10 proteins ranked by betweenness centrality. Of the five proteins, BCL2, JUN, and MAPK3 were found to be involved in more than five of 12 KEGG pathways related with encephalitis. The KEGG pathways that included all three proteins were estrogen signaling pathway, apoptosis, and neurotrophin signaling pathway. Meanwhile, there are various studies about that BCL2, JUN, and MAPK3 are associated with neuroinflammation, neurodegeneration and cognitive deficits. BCL2, generally recognized as anti-apoptotic protein, could mediate neuronal differentiation by regulating lipopolysaccharide-induced inflammation in cortical neural stem cells, suggesting a protective effect of BCL2 in neuroinflammation (Park and Han, 2022). c-Jun, a product of JUN gene, plays a complex role by regulating neurodegeneration, neuroinflammation, neuronal regeneration, and neuroplasticity (Raivich, 2008). Similarly, MAPK3, also known as ERK1, is essential for normal physiological brain function such as learning and memory, but it also involved in neuroinflammation and neurodegeneration (Impey et al., 1999; Peng et al., 2010; Sun and Nan, 2017). Therefore, dysregulating the three key genes, BCL2, JUN, and MAPK3, may induce alternation in neuronal microenvironment, ultimately leading to cognitive deficits.
In conclusion, we confirmed the protective effects of AR extract, which attenuated neuronal damage in a CYP-induced cognitive impairment mouse model. Furthermore, based on the network pharmacology analysis, AR compounds may exert their neuroprotective effects against CYP-induced neuroinflammation through key candidate target genes, BCL2, JUN, and MAPK3, although further validation is required to confirm these targets and their roles.
Acknowledgments
This work was supported by a grant from the Jeju Institute of Korean Medicine (JIKOM-C-230004), Korea Institute of Oriental Medicine (ERT2011180 and KSN2511030), Convergence Research Group Project of the National Research Council of Science (CRC21021) and Regional Innovation System & Education (RISE) through the Gwangju RISE Center, funded by the Ministry of Education (MOE) and the Gwangju Metropolitan Government (20250527000003426624).
References
-
Ahles TA and Saykin A. (2001). Cognitive effects of standard-dose chemotherapy in patients with cancer. Cancer Investigation. 19:812-820.
[https://doi.org/10.1081/CNV-100107743]

-
Bagnall-Moreau C, Chaudhry S, Salas-Ramirez K, Ahles T and Hubbard K. (2019). Chemotherapy-induced cognitive impairment is associated with increased inflammation and oxidative damage in the hippocampus. Molecular Neurobiology. 56:7159-7172.
[https://doi.org/10.1007/s12035-019-1589-z]

-
Cao P, Xie P, Wang X, Wang J, Wei J and Kang WY. (2017). Chemical constituents and coagulation activity of Agastache rugosa. BMC Complementary and Alternative Medicine. 17:93. https://bmccomplementmedtherapies.biomedcentral.com/articles/10.1186/s12906-017-1592-8, (cited by 2025 Aug. 10).
[https://doi.org/10.1186/s12906-017-1592-8]

-
Chen L, Zhu T, Qi J, Zhang Y, Zhang Z and Liu H. (2021). Pharmacological mechanism of JiaWeiSiWu granule in the treatment of hypertension based on network pharmacology. Annals of Palliative Medicine. 10:7486-7513.
[https://doi.org/10.21037/apm-21-1140]

-
de Jonge ME, Huitema ADR, Rodenhuis S and Beijnen JH. (2005). Clinical pharmacokinetics of cyclophosphamide. Clinical Pharmacokinetics. 44:1135-1164.
[https://doi.org/10.2165/00003088-200544110-00003]

-
de Sousa MAR, dos Santos Teixeira G, Marquesa RB, de Sousa LMR, Ramos RM, de França Bento RR, Neto BCV, Gusmão SBS, Sá JLS and Maia Filho ALM. (2024). Therapeutic actions of methyl eugenol in acute lung inflammation induced in rats. South African Journal of Botany. 169:341-349.
[https://doi.org/10.1016/j.sajb.2024.04.023]

-
Desta KT, Kim GS, Kim YH, Lee WS, Lee SJ, Jin JS, Abd El-Aty AM, Shin HC, Shim JH and Shin SC. (2016). The polyphenolic profiles and antioxidant effects of Agastache rugosa Kuntze(Banga) flower, leaf, stem and root. Biomedical Chromatography. 30:225-231.
[https://doi.org/10.1002/bmc.3539]

-
Eddin LB, Jha NK, Meeran MFN, Kesari KK, Beiram R and Ojha S. (2021). Neuroprotective potential of limonene and limonene containing natural products. Molecules. 26:4535. https://www.mdpi.com/1420-3049/26/15/4535, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/molecules26154535]

-
Gao K, Zhu Y, Wang H, Gong X, Yue Z, Lv A and Zhou X. (2021). Network pharmacology reveals the potential mechanism of Baiying Qinghou decoction in treating laryngeal squamous cell carcinoma. Aging (Albany NY), 13:26003-26021. https://www.aging-us.com/article/203786/text, (cited by 2025 Aug. 10).
[https://doi.org/10.18632/aging.203786]

-
Gogoi R, Loying R, Sarma N, Begum T, Pandey SK and Lal M. (2020). Comparative analysis of in-vitro biological activities of methyl eugenol rich Cymbopogon khasianus Hack., leaf essential oil with pure methyl eugenol compound. Current Pharmaceutical Biotechnology. 21:927-938.
[https://doi.org/10.2174/1389201021666200217113921]

-
Hirshman NA, Hughes FM, Jr., Jin H, Harrison WT, White SW, Doan I, Harper SN, Leidig PD and Purves JT. (2020). Cyclophosphamide-induced cystitis results in NLRP3-mediated inflammation in the hippocampus and symptoms of depression in rats. American Journal of Physiology: Renal Physiology. 318:F354-F362.
[https://doi.org/10.1152/ajprenal.00408.2019]

-
Hodgson KD, Hutchinson AD, Wilson CJ and Nettelbeck T. (2013). A meta-analysis of the effects of chemotherapy on cognition in patients with cancer. Cancer Treatment Reviews. 39:297-304.
[https://doi.org/10.1016/j.ctrv.2012.11.001]

-
Hussein Z, Michel HE, El-Naga RN, El-Demerdash E and Mantawy EM. (2024). Coenzyme Q10 ameliorates cyclophosphamide-induced chemobrain by repressing neuronal apoptosis and preserving hippocampal neurogenesis: Mechanistic roles of Wnt/β-catenin signaling pathway. Neurotoxicology. 105:21-33.
[https://doi.org/10.1016/j.neuro.2024.08.003]

-
Ibrahim KM, Darwish SF, Mantawy EM and El-Demerdash E. (2024). Molecular mechanisms underlying cyclophosphamideinduced cognitive impairment and strategies for neuroprotection in preclinical models. Molecular and Cellular Biochemistry. 479:1873-1893.
[https://doi.org/10.1007/s11010-023-04805-0]

-
Impey S, Obrietan K and Storm DR. (1999). Making new connections: Role of ERK/MAP kinase signaling in neuronal plasticity. Neuron. 23:11-14.
[https://doi.org/10.1016/S0896-6273(00)80747-3]

-
Kang S, Lee N, Jung B, Jeong H, Moon C, Park SI, Yun S, Yim T, Oh JM, Kim JW, Song JH, Chae S and Kim JS. (2024). Anti-amnesic effect of Agastache rugosa on scopolamineinduced memory impairment in mice. Pharmaceuticals. 17:1173. https://www.mdpi.com/1424-8247/17/9/1173, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/ph17091173]

-
Ko SJ, Han G, Kim SK, Seo JG, Chung WS, Ryu B, Kim J, Yeo I, Lee BJ, Lee JM and Park JW. (2013). Effect of korean herbal medicine combined with a probiotic mixture on diarrhea-dominant irritable bowel syndrome: A double-blind, randomized, placebo-controlled trial. Evidence-Based Complementary and Alternative Medicine. 2013:824605.
[https://doi.org/10.1155/2013/824605]

-
Lange M, Giffard B, Noal S, Rigal O, Kurtz JE, Heutte N, Lévy C, Allouache D, Rieux C, Le Fel J, Daireaux A, Clarisse B, Veyret C, Barthélémy P, Longato N, Eustache F and Joly F. (2014). Baseline cognitive functions among elderly patients with localised breast cancer. European Journal of Cancer. 50:2181-2189.
[https://doi.org/10.1016/j.ejca.2014.05.026]

-
Lee AY, Lee JY and Chun JM. (2020). Exploring the mechanism of Gyejibokryeong-hwan against atherosclerosis using network pharmacology and molecular docking. Plants. 9:1750. https://www.mdpi.com/2223-7747/9/12/1750, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/plants9121750]

-
Lee S, Ryu SM, Kim DH, Lee YE, Lee SJ, Kang S, Kim JS and Lee SI. (2022). Neuroprotective effect of Geijigadaehwang-tang against trimethyltin-induced hippocampal neurodegeneration: An in vitro and in vivo study. Journal of Ethnopharmacology. 296:115451.
[https://doi.org/10.1016/j.jep.2022.115451]

-
Matsos A and Johnston IN. (2019). Chemotherapy-induced cognitive impairments: A systematic review of the animal literature. Neuroscience and Biobehavioral Reviews. 102:382-399.
[https://doi.org/10.1016/j.neubiorev.2019.05.001]

-
Monje ML, Toda H and Palmer TD. (2003). Inflammatory blockade restores adult hippocampal neurogenesis. Science. 302:1760-1765.
[https://doi.org/10.1126/science.1088417]

-
Nam HH, Kim JS, Lee J, Seo YH, Kim HS, Ryu SM, Choi G, Moon BC and Lee AY. (2020). Pharmacological effects of Agastache rugosa against gastritis using a network pharmacology approach. Biomolecules. 10:1298. https://www.mdpi.com/2218-273X/10/9/1298, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/biom10091298]

-
Park SW, Lee AY, Lim JO, Lee SJ, Kim WI, Yang YG, Kim B, Kim JS, Chae SW, Na K, Seo YS and Shin IS. (2022). Loranthus tanakae Franch. & Sav. suppresses inflammatory response in cigarette smoke condensate exposed bronchial epithelial cells and mice. Antioxidants. 11:1885. https://www.mdpi.com/2076-3921/11/10/1885, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/antiox11101885]

-
Park SY and Han JS. (2022). Neuroprotective effect of Bcl-2 on lipopolysaccharide-induced neuroinflammation in cortical neural stem cells. International Journal of Molecular Sciences. 23:6399.
[https://doi.org/10.3390/ijms23126399]

-
Park YJ, Yang HJ, Li W, Oh YC and Go Y. (2022). Menthae herba attenuates neuroinflammation by regulating CREB/Nrf2/HO-1 pathway in BV2 microglial cells. Antioxidants. 11:649. https://www.mdpi.com/2076-3921/11/4/649, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/antiox11040649]

-
Peng S, Zhang Y, Zhang J, Wang H and Ren B. (2010). ERK in learning and memory: A review of recent research. International Journal of Molecular Sciences. 11:222-232.
[https://doi.org/10.3390/ijms11010222]

-
Raivich G. (2008). c-Jun expression, activation and function in neural cell death, inflammation and repair. Journal of Neurochemistry. 107:898-906.
[https://doi.org/10.1111/j.1471-4159.2008.05684.x]

-
Rinik UR, Kim JE, Lee E, Kwon O and Jung BH. (2024). Development of simultaneous quantitative analytical method for three active components of Korean mint (Agastache rugosa (Fisch. & C.A.Mey.) Kuntze) extract in human plasma using ultra-high-performance liquid chromatography-tandem mass spectrometry. Journal of Chromatography. B: Analytical Technologies in the Biomedical and Life Sciences. 1232:123957.
[https://doi.org/10.1016/j.jchromb.2023.123957]

-
Roy A, Park H-J, Abdul QA, Jung HA and Choi JS. (2018). Pulegone exhibits anti-inflammatory activities through the regulation of NF-κB and Nrf-2 signaling pathways in LPS-stimulated RAW 264.7 cells. Natural Product Sciences. 24:28-35.
[https://doi.org/10.20307/nps.2018.24.1.28]

-
Sato K. (2015). Effects of microglia on neurogenesis. Glia. 63:1394-1405.
[https://doi.org/10.1002/glia.22858]

-
Sekeres MJ, Bradley-Garcia M, Martinez-Canabal A and Winocur G. (2021). Chemotherapy-induced cognitive impairment and hippocampal neurogenesis: A review of physiological mechanisms and interventions. International Journal of Molecular Sciences. 22:12697.
[https://doi.org/10.3390/ijms222312697]

-
Seo YS, Ryu SM, Lee J, Jeong H, Choi G, Moon BC, Lim JO, Nam HH, Kim JS and Lee S. (2025). Protective effect of cast-off skin of Cicadidae Periostracum water extract in a radiation-induced testicular injury mice model. Food Science & Nutrition. 13:e70198.
[https://doi.org/10.1002/fsn3.70198]

-
Shin S. (2004). Essential oil compounds from Agastache rugosa as antifungal agents against Trichophyton species. Archives of Pharmacal Research. 27:295-299.
[https://doi.org/10.1007/BF02980063]

-
Silberfarb PM. (1983). Chemotherapy and cognitive defects in cancer patients. Annual Review of Medicine. 34:35-46.
[https://doi.org/10.1146/annurev.me.34.020183.000343]

-
Song JH, Nam HH, Park I, Yang S, Chun JM, Seo YS, Kim HY, Moon BC, Kang S, Moon C, Kang SI, Song JH and Kim JS. (2024). Comparative morphology of island and inland Agastache rugosa and their gastroprotective effects in EtOH/HCl-induced gastric mucosal gastritis. Planta Medica. 90:4-12.
[https://doi.org/10.1055/a-2189-7272]

-
Sun J and Nan G. (2017). The extracellular signal-regulated kinase 1/2 pathway in neurological diseases: A potential therapeutic target(Review). International Journal of Molecular Medicine. 39:1338-1346.
[https://doi.org/10.3892/ijmm.2017.2962]

-
Wang ZJ and Heinbockel T. (2018). Essential oils and their constituents targeting the GABAergic system and sodium channels as treatment of neurological diseases. Molecules. 23:1061. https://www.mdpi.com/1420-3049/23/5/1061, (cited by 2025 Aug. 10).
[https://doi.org/10.3390/molecules23051061]

-
Wei X, Hou W, Liang J, Fang P, Dou B, Wang Z, Sai J, Xu T, Ma C, Zhang Q, Cheng F, Wang X and Wang Q. (2021). Network pharmacology-based analysis on the potential biological mechanisms of sinisan against non-alcoholic fatty liver disease. Frontiers in Pharmacology. 12:693701. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2021.693701/full, (cited by 2025 Aug. 10).
[https://doi.org/10.3389/fphar.2021.693701]

-
Xu J, Kang F, Wang W, Liu S, Xie J and Yang X. (2022). Comparison between heat-clearing medicine and antirheumatic medicine in treatment of gastric cancer based on network pharmacology, molecular docking, and tumor immune infiltration analysis. Evidence-Based Complementary and Alternative Medicine. 2022:7490279.
[https://doi.org/10.1155/2022/7490279]

-
Zielińska S and Matkowski A. (2014). Phytochemistry and bioactivity of aromatic and medicinal plants from the genus Agastache(Lamiaceae). Phytochemistry Reviews. 13:391-416.
[https://doi.org/10.1007/s11101-014-9349-1]